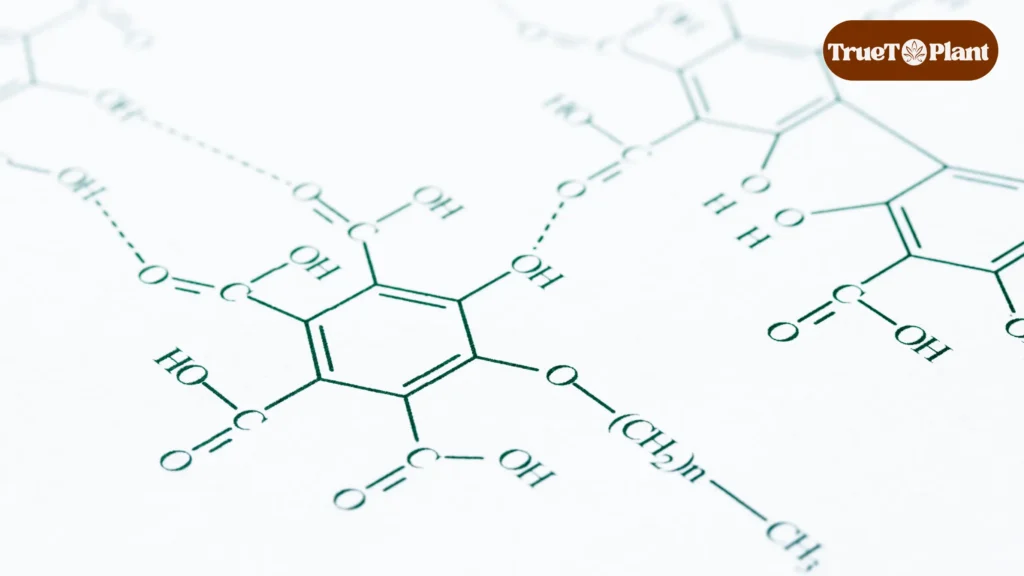
While cannabinoids dominate cannabis research, flavonoids quietly shape the cultivar-specific effects consumers experience. These secondary metabolites, derived from flavone with powerful antioxidant properties and formed through plant metabolite formation, represent over 10,000 identified molecules across plant biology. In cannabis, unique compounds like cannflavins deliver anti-inflammatory effects up to 30 times more potent than aspirin.
Different cultivars produce distinct flavonoid profiles that influence therapeutic outcomes beyond cannabinoid content alone. Understanding the biosynthetic pathways behind these compounds unlocks targeted breeding strategies for specific effects. Yet most cultivation programs overlook flavonoid synthesis entirely, focusing exclusively on THC and CBD levels. This article examines the genetic mechanisms controlling flavonoid production in cannabis, revealing how metabolic profiling and data-driven insights can optimize cultivar development for predictable, reproducible experiences.
What Are Flavonoids in Cannabis? Beyond Terpenes and Cannabinoids
Flavonoids are a class of secondary metabolites responsible for pigmentation and physiological defense mechanisms across plant species. In cannabis, these compounds work alongside cannabinoids to produce cultivar-specific effects, yet they operate through entirely different mechanisms than their better-known counterparts.
What is the difference between terpenes and flavonoids in cannabis?
Terpenes are volatile aromatic compounds that evaporate easily and directly activate cannabinoid receptors, while flavonoids are non-volatile pigment molecules that provide antioxidant and anti-inflammatory benefits through oxidative stress modulation. Terpenes deliver scent profiles and receptor activity. Flavonoids contribute color expression and UV protection while influencing therapeutic outcomes through distinct biosynthetic pathways.
Both compound classes participate in the entourage effect, but flavonoids remain stable under conditions where terpenes volatilize. This stability makes flavonoid composition particularly valuable for understanding consistent therapeutic profiles across different consumption methods.
In plant biology, flavonoids serve foundational roles beyond aesthetics. They regulate oxidative stress responses, protect against UV radiation, and participate in cellular signaling networks. Cannabis produces unique flavonoids like cannflavins alongside common anthocyanins that determine purple coloration in certain cultivars.
The distinction matters for breeding programs targeting specific effects. While terpene profiles receive significant attention, flavonoid synthesis pathways offer untapped potential for developing cultivars with predictable, scientifically validated benefits. Metabolic profiling platforms can now track both compound classes simultaneously, enabling data-driven cultivation strategies that optimize the full spectrum of bioactive molecules.
Cannflavins: The Cannabis-Exclusive Flavonoids
Which flavonoid unique to cannabis has potent anti-inflammatory effects?
Cannflavin A stands out as cannabis’s most powerful anti-inflammatory flavonoid, demonstrating efficacy 30 times greater than aspirin in oxidative stress modulation. This prenylflavonoid exists exclusively in cannabis alongside cannflavins B and C, forming a trio of compounds found nowhere else in nature.
Cannflavins A
Cannflavin A delivers neuroprotective properties through distinct metabolic pathways separate from cannabinoid receptor activation. Recent 2024 clinical trials demonstrate that full-spectrum products containing cannflavins enhance analgesic effects beyond isolated cannabinoid formulations. The phenolic profile of these molecules suggests biomedical applications extending well past traditional pain management.
Cannflavin B contributes similar therapeutic potential, though research indicates slightly reduced potency compared to its A counterpart. Cannflavin C remains less studied but shares the prenylflavonoid structure characteristic of this cannabis-exclusive class. Together, when ingested orally, these compounds participate in the entourage effect alongside beta-caryophyllene’s effects and other secondary metabolites.
Stellenbogh University research reveals that cannflavin biosynthesis follows specialized gene expression patterns unique to cannabis genetics. Understanding these pathways enables breeding programs to select for elevated cannflavin production, optimizing cultivars for specific therapeutic outcomes. Yet quantifying cannflavin content requires advanced metabolic profiling platforms – most standard testing protocols measure only cannabinoid concentrations, overlooking these bioactive flavonoids entirely.
What Aspect of the Cannabis Plant Is Affected by Flavonoids?
Flavonoids influence multiple plant systems simultaneously, creating the visible and invisible characteristics that define cultivar identity. Anthocyanin synthesis drives purple pigmentation in cultivars through pH-dependent color expression, while other flavonoid subclasses produce yellow and red hues across different genetic backgrounds. This pigment production extends beyond aesthetics – these compounds absorb harmful UV radiation, protecting cellular DNA from photodamage during outdoor cultivation.
Defense mechanisms represent another critical function. Flavonoids demonstrate antimicrobial activity against pathogens and deter herbivorous pests through astringent taste compounds. Research confirms these secondary metabolites modulate oxidative stress responses, enabling plants to survive environmental challenges that would otherwise compromise growth. The antioxidant capacity varies significantly between cultivars based on genetic regulation of biosynthetic pathways.
Sensory profiles receive direct input from flavonoid composition. Recent consumer trend data shows 68% of users associate flavonoid-rich cultivars with more complex taste experiences, including bitterness and astringency that complement terpenes and cannabinoids. These flavor contributions persist through processing where volatile compounds evaporate.
Therapeutic applications emerge from flavonoid interactions with human physiology. Anti-inflammatory and neuroprotective properties operate through mechanisms distinct from cannabinoid receptor pathways, contributing to entourage effects in full-spectrum formulations. Glycosylation processes stabilize anthocyanin molecules, ensuring consistent bioavailability across consumption methods.
Understanding these multifaceted roles enables breeding programs to target specific outcomes through metabolic profiling. Cultivar-specific flavonoid expression determines not just appearance, but resilience, flavor complexity, and functional compound delivery.
The Flavonoid Biosynthesis Pathway: From Phenylalanine to Pigments
Flavonoid synthesis begins with phenylalanine, an amino acid that enters the phenylpropanoid pathway through phenylalanine ammonia-lyase (PAL) enzyme activity. This upstream conversion generates p-Coumaroyl-CoA, which then combines with malonyl-CoA through chalcone synthase (CHS) – the gateway enzyme controlling flavonoid production. CHS catalyzes chalcone formation, creating the foundational scaffold from which all flavonoid subclasses diverge.
These biosynthetic events mirror the broader processes involved in how plants create complex chemistry, where metabolic pathways branch into numerous bioactive compounds.
From chalcone, the pathway branches into distinct routes producing different compound families. Flavonoid 3-hydroxylase (F3H) directs substrates toward anthocyanins synthesis, generating the pigmentation molecules responsible for purple and red coloration in cannabis cultivars. Alternative branches produce flavones through flavone synthase (FNS) activity, while flavonol synthase (FLS) generates flavonols like quercetin and kaempferol with documented neuroprotective properties.
Genetic regulation determines which branches dominate in specific cultivars. Transcriptome analyses reveal that gene expression patterns controlling CHS, F3H, and downstream enzymes vary dramatically between chemotypes. CRISPR-Cas9 applications now enable precision breeding programs to upregulate specific enzyme genes, enhancing targeted flavonoid accumulation without compromising cannabinoid production.
Environmental factors modulate this genetic blueprint. UV exposure increases PAL activity, elevating total flavonoid content as plants respond to photostress. Temperature fluctuations affect anthocyanidin reductase expression, explaining why purple cultivars display more intense pigmentation during cooler flowering periods.
Metabolic profiling platforms track these biosynthetic outputs across growth stages, revealing how cannabis aroma compounds and flavonoids develop simultaneously through parallel pathways. Understanding enzyme kinetics and regulatory networks enables cultivators to optimize conditions for desired flavonoid profiles, moving beyond trial-and-error approaches toward data-driven cultivation strategies.
Anthocyanin Synthesis: The Genetics Behind Purple Cannabis
Anthocyanin accumulation in cannabis follows a genetically controlled branch of the flavonoid pathway, where dihydroflavonol reductase (DFR) and anthocyanidin synthase (ANS) convert colorless precursors into vibrant pigments. These enzymes operate downstream from the chalcone scaffold, directing metabolite flow specifically toward purple and blue coloration rather than alternative flavonoid subclasses.
Cultivars like Granddaddy Purple carry genetic markers that upregulate DFR expression, enabling high anthocyanin concentrations absent in green phenotypes. Anthocyanidin reductase (ANR) further modifies these pigments into stable proanthocyanidins, which persist through drying and processing. Recent genetic diversity analyses reveal that functional insertions and deletions (InDels) within regulatory regions determine whether plants activate this pigmentation cascade under appropriate conditions.
Temperature triggers anthocyanin expression through stress-response pathways. Recent 2024 research confirms that purple cultivars display peak pigmentation when nighttime temperatures drop below 15°C during flowering, activating transcription factors that boost ANS activity. Warmer conditions suppress these genetic switches even in genetically predisposed plants, explaining why identical clones produce variable coloration across different environments.
pH levels influence anthocyanin color expression post-synthesis. Acidic cellular conditions shift pigments toward red hues, while neutral to alkaline pH produces blue and purple tones. This pH-dependent chromatic variation operates independently of genetic anthocyanin quantity, adding complexity to breeding programs targeting specific visual outcomes.
For breeders, selecting parent lines with confirmed DFR and ANS expression patterns enables predictable purple offspring. Metabolic profiling platforms now quantify anthocyanin concentrations alongside flavonoid synthesis markers, providing data-driven selection criteria beyond visual phenotype assessment. Understanding these genetic controls transforms color development from unpredictable trait to reproducible breeding target.
Genetic Mapping and Cultivar-Specific Flavonoid Profiles
Whole-genome sequencing reveals how chromosomal locations control flavonoid biosynthesis across cannabis cultivars. Quantitative trait loci (QTL) mapping identifies specific gene clusters responsible for cannflavin production, anthocyanin accumulation, and flavonol expression. These genomic regions vary between chemotypes, explaining why CBD-dominant cultivars produce different secondary metabolite signatures than THC-rich varieties even when grown under identical conditions.
Multi-omics integration combines transcriptomics, proteomics, and metabolomics data to map gene expression patterns onto chemical outputs. Research demonstrates that cultivars with elevated cannflavin A concentrations carry specific allelic variants in prenyltransferase genes absent from low-producing lines. These genetic markers enable molecular-assisted breeding, where DNA screening replaces time-intensive chemical analysis during parent selection.
CRISPR-Cas9 applications now target regulatory sequences controlling flavonoid pathway flux. Recent metabolic engineering achieved 40-fold increases in precursor molecules through precision gene editing, demonstrating scalability for optimizing therapeutic compound ratios. This technology allows breeders to enhance cannflavin synthesis without disrupting cannabinoid production pathways that share upstream precursors.
High-performance liquid chromatography coupled with mass spectrometry (HPLC-MS) provides the analytical precision necessary for validating genetic predictions. Quantifying flavonoid concentrations below 0.1% dry weight requires sensitive detection platforms that track dozens of compounds simultaneously across developmental stages.
For personalized medicine applications, cultivar-specific metabolic fingerprints offer reproducible therapeutic profiles. Genetic mapping transforms breeding from phenotype observation into predictable molecular design, where targeted gene expression produces consistent flavonoid signatures matched to clinical outcomes.
The Entourage Effect: How Flavonoids Enhance Cannabinoid Activity
Cannabinoid bioavailability increases measurably when flavonoids participate in delivery mechanisms. Recent metabolomic analyses demonstrate that flavonoid-rich extracts enhance cannabinoid absorption and endocannabinoid system modulation, creating therapeutic outcomes unattainable through isolated compounds. Quercetin and kaempferol improve membrane permeability, facilitating cannabinoid transport across cellular barriers while providing independent antioxidant activity that protects against oxidative stress.
Chemotype-specific research reveals how cultivar flavonoid profiles determine therapeutic differentiation beyond cannabinoid ratios alone. High-cannflavin cultivars deliver superior anti-inflammatory responses compared to chemically identical cannabinoid formulations lacking these prenylflavonoids. Anthocyanin-rich purple varieties demonstrate enhanced neuroprotective properties through synergistic interactions with THC and CBD, operating via complementary molecular pathways that amplify clinical benefits.
Pharmaceutical standardization efforts confirm that full-spectrum formulations preserving native flavonoid content produce reproducible patient experiences superior to isolated cannabinoid products. This molecular synergy explains why whole-plant extracts outperform synthetic alternatives in clinical applications, despite identical cannabinoid concentrations.
For formulation development, maintaining flavonoid integrity requires technology that captures heat-sensitive compounds alongside volatile terpenes. Entour’s True To Plant® platform analyzes over 200 bioactive molecules including flavonoid markers, enabling precise cultivar replication that preserves the complete phytochemical architecture responsible for strain-specific effects.
Engineering the Future of Cannabis Chemistry
Precision breeding transforms cultivar development from observational selection into molecular design. Mapping flavonoid biosynthesis genes enables breeders to predict purple phenotypes, cannflavin concentrations, and therapeutic profiles before plants reach maturity. Synthetic biology tools now optimize enzyme expression ratios, creating chemotypes with reproducible flavonoid signatures matched to clinical applications.
Authentic cultivar replication demands comprehensive metabolic fingerprinting beyond cannabinoid percentages. Entour’s platform considers flavonoid markers alongside volatile compounds, preserving the complete phytochemical architecture that defines strain identity. This analytical depth ensures formulations deliver consistent experiences rooted in cultivar-specific chemistry rather than approximations.
Personalized medicine applications require standardized flavonoid profiles validated through multi-omics integration. Recent advances in metabolomics reveal how individual patients respond differently to quercetin-rich versus cannflavin-dominant chemotypes, enabling targeted therapeutic matching. Genetic innovation accelerates this precision through CRISPR-enhanced cultivars designed for specific bioactive ratios.
The future belongs to formulations honoring botanical complexity. Investing in genetic mapping, metabolic profiling platforms, and flavor authenticity creates products where science meets sensory experience. Understanding flavonoid synthesis pathways unlocks cannabis chemistry’s full therapeutic potential – moving the industry toward data-driven innovation grounded in molecular truth.